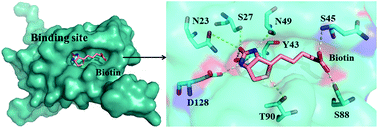
Entropic effect and residue specific entropic contribution to the cooperativity in streptavidin–biotin binding - Nanoscale (RSC Publishing)

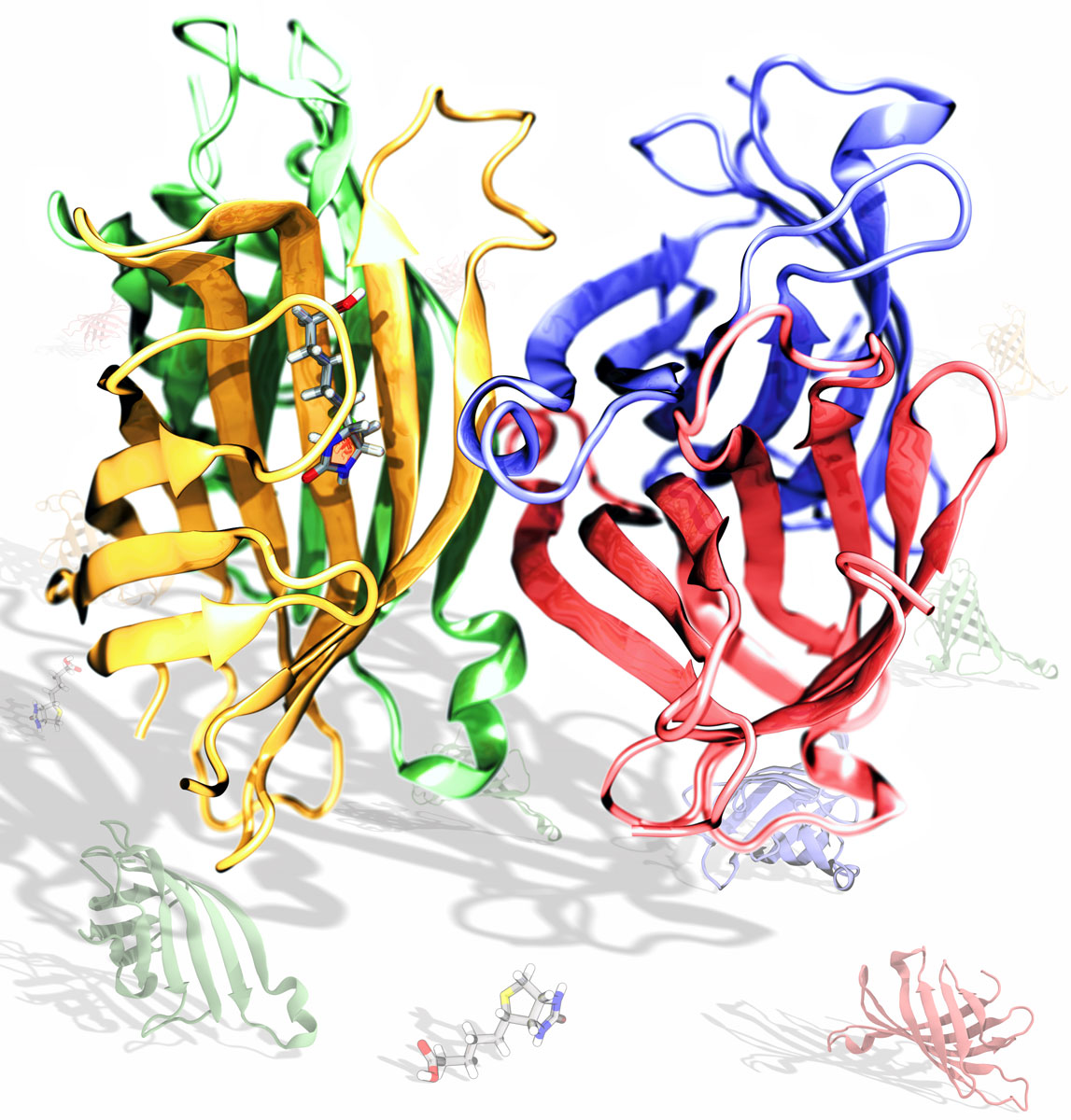
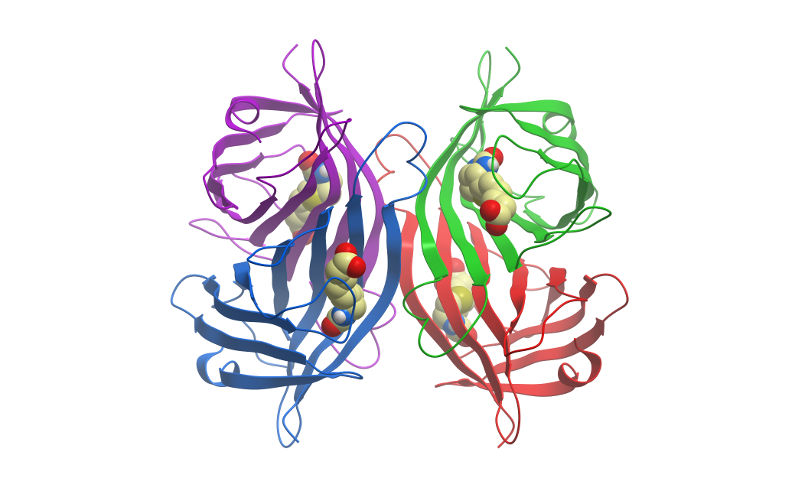
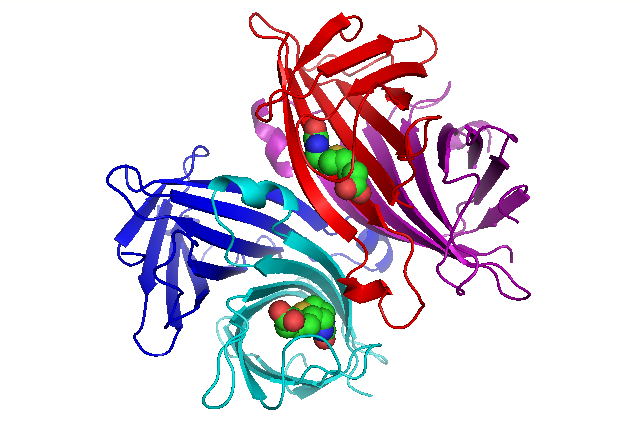
The origin of the cooperativity in the streptavidin-biotin system: A computational investigation through molecular dynamics simulations | Scientific Reports
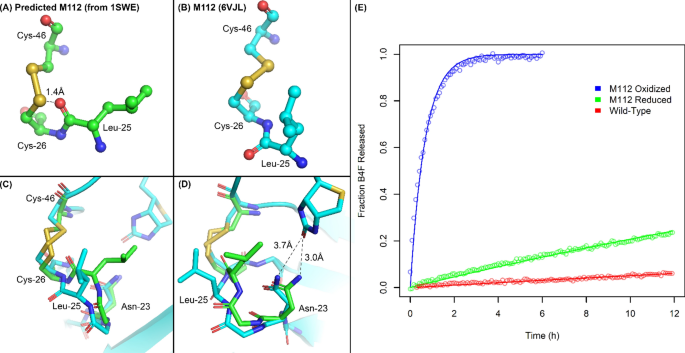
Engineering a disulfide-gated switch in streptavidin enables reversible binding without sacrificing binding affinity | Scientific Reports

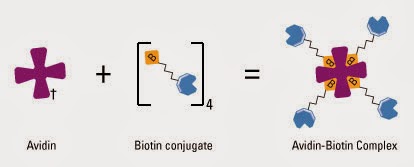
a) Avidin/biotin and streptavidin/biotin complex formation. For the... | Download Scientific Diagram
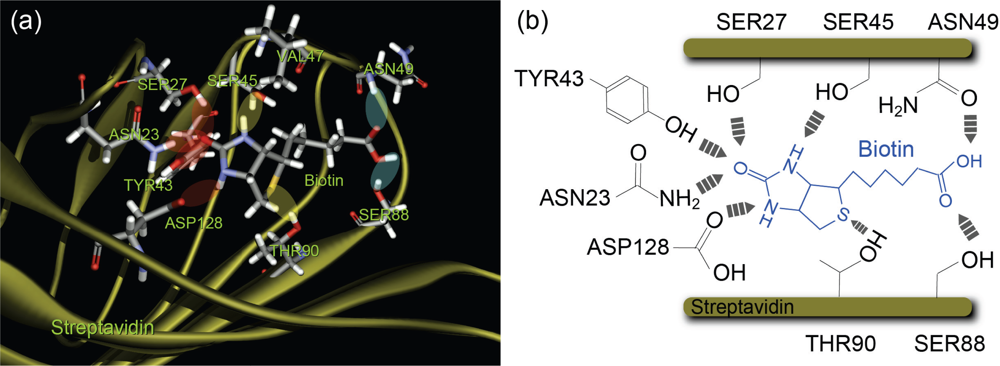
IJMS | Free Full-Text | Reconsideration of Dynamic Force Spectroscopy Analysis of Streptavidin-Biotin Interactions

Hidden variety of biotin-streptavidin/avidin local interactions revealed by site-selective dynamic force spectroscopy. | Semantic Scholar
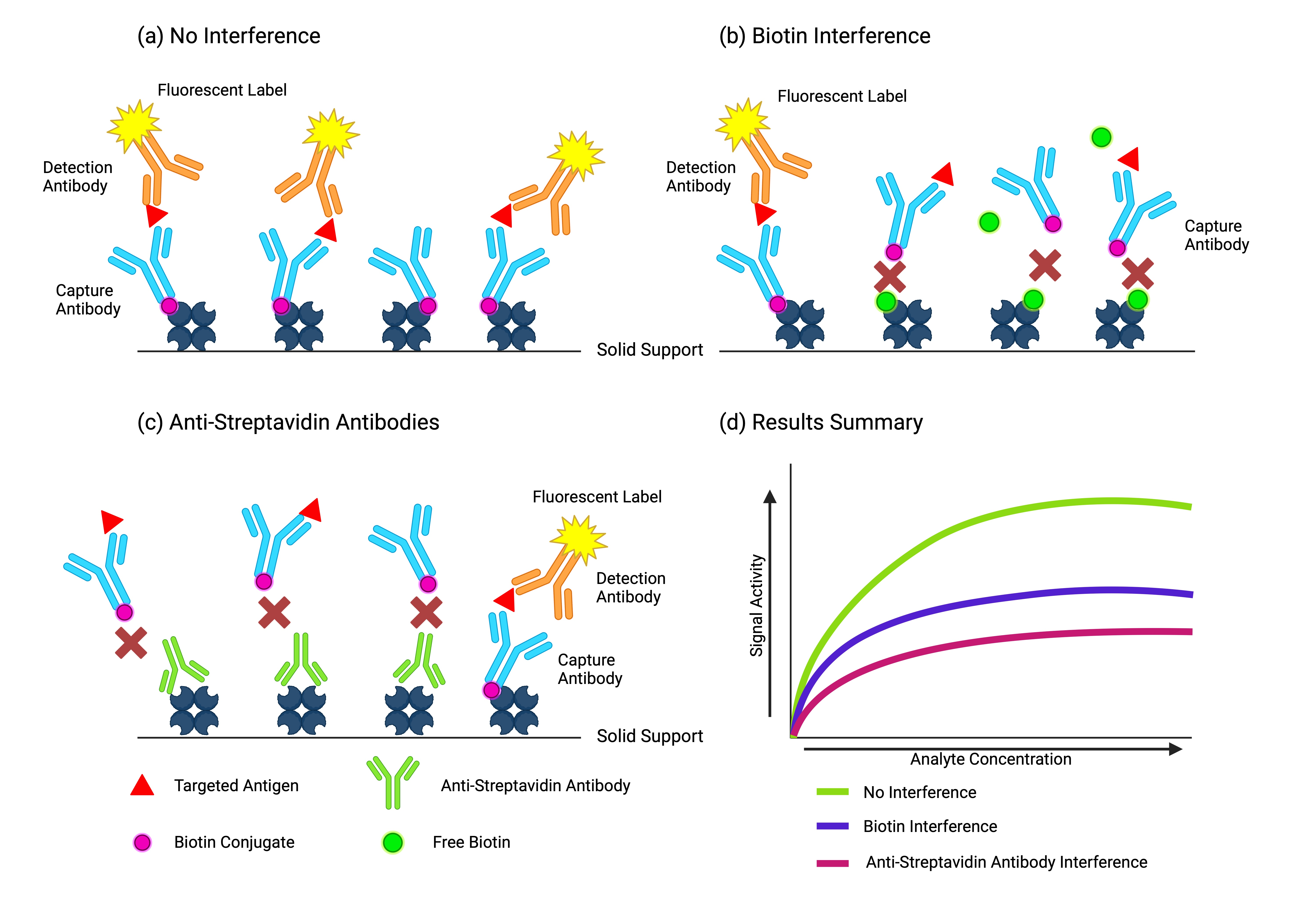
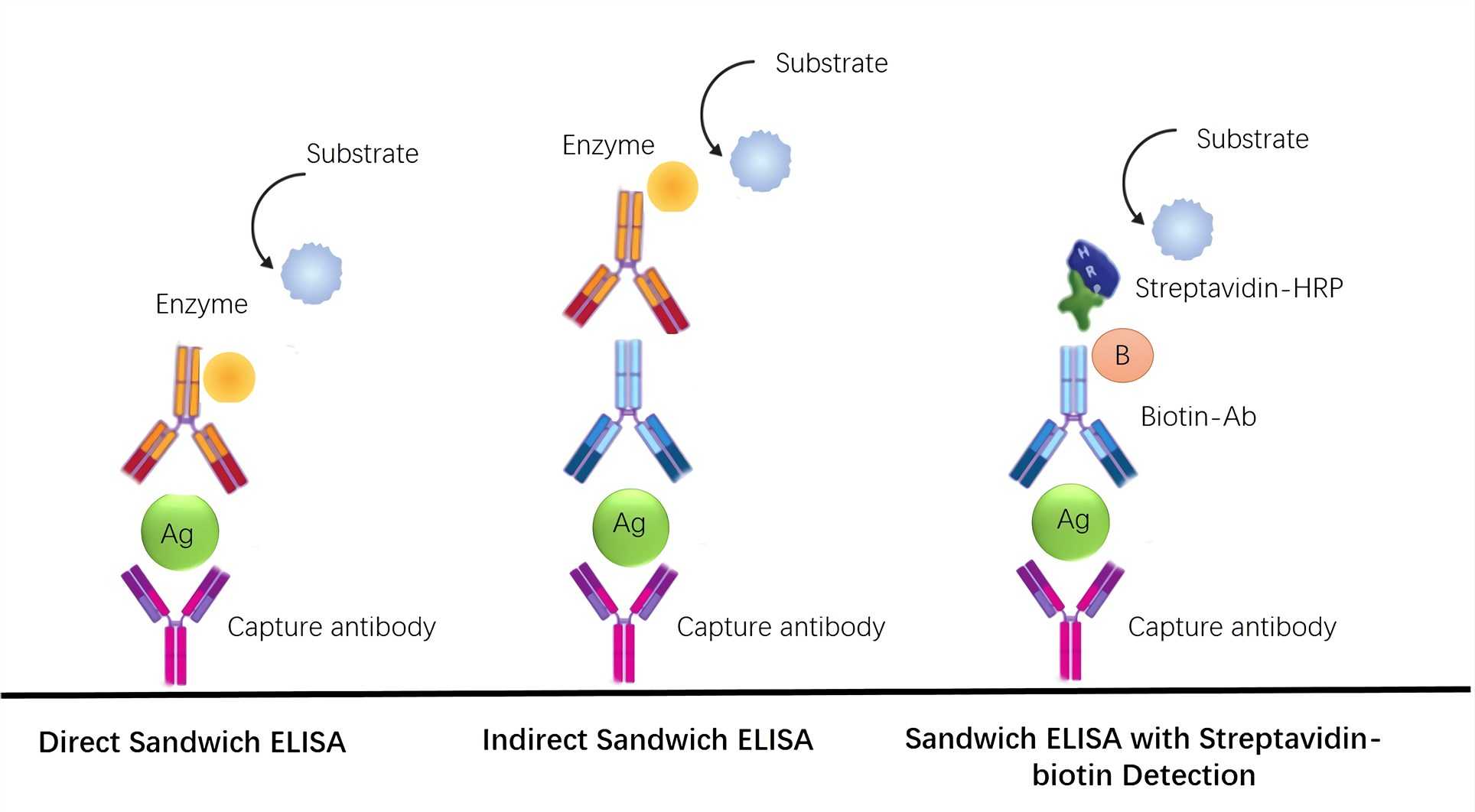
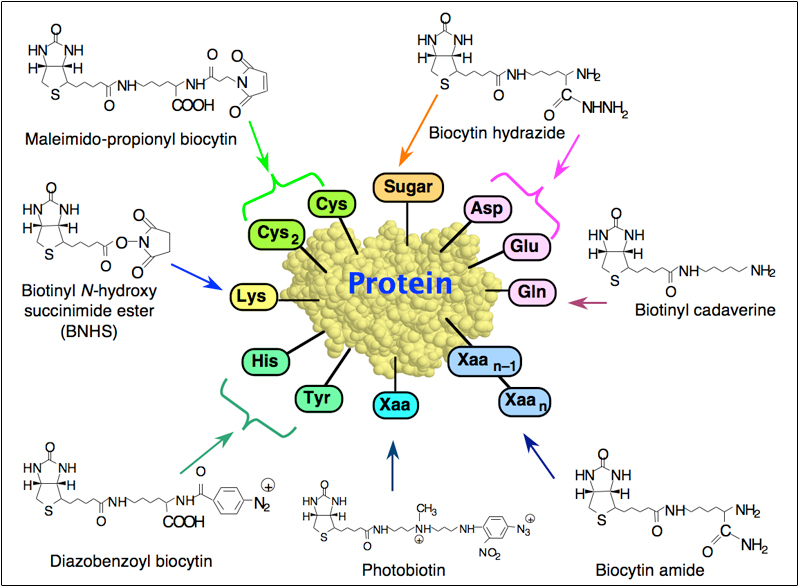
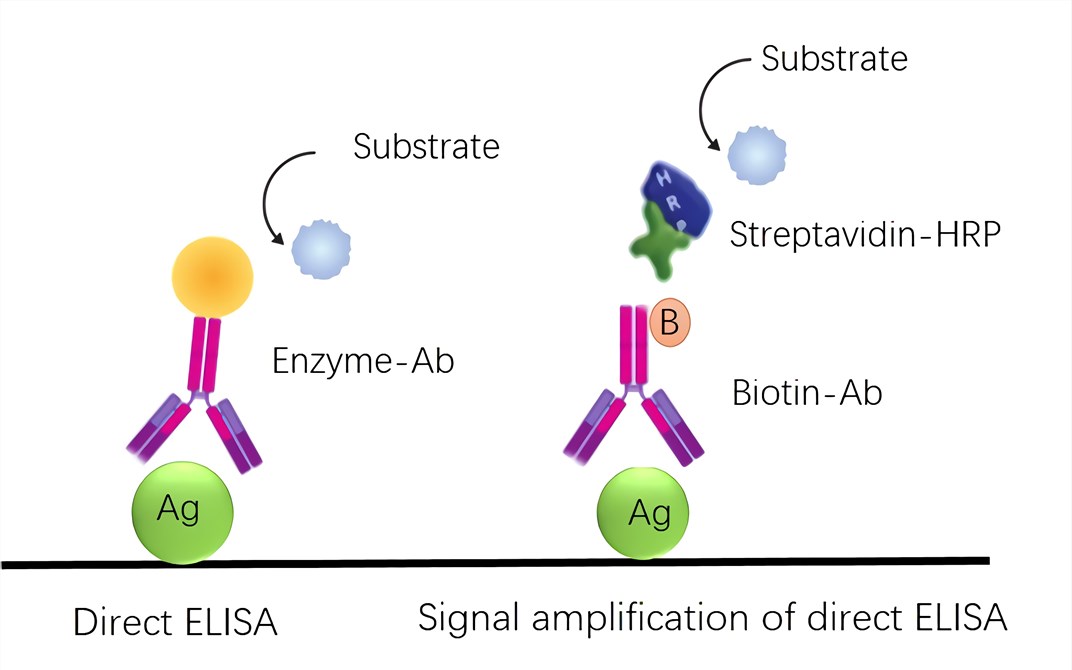
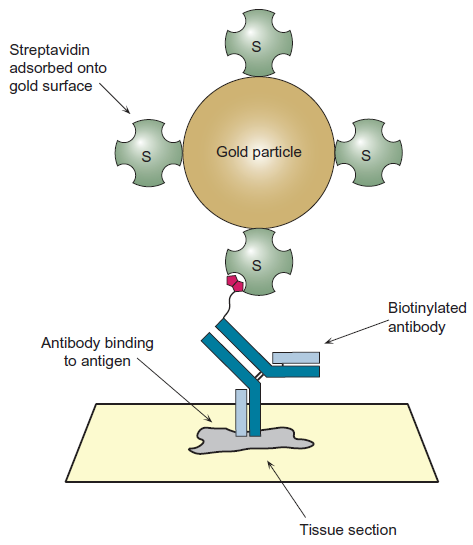
CIMB | Free Full-Text | An Analysis of the Biotin–(Strept)avidin System in Immunoassays: Interference and Mitigation Strategies

Purification of DNA‐Binding Proteins Using Biotin/Streptavidin Affinity Systems - Chodosh - 1996 - Current Protocols in Molecular Biology - Wiley Online Library

Monitoring of Real-Time Streptavidin−Biotin Binding Kinetics Using Droplet Microfluidics | Analytical Chemistry
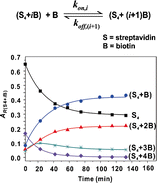
Dissociation Kinetics of the Streptavidin–Biotin Interaction Measured Using Direct Electrospray Ionization Mass Spectrometry Analysis | Journal of The American Society for Mass Spectrometry